Baseline characteristics of study patients following ESD procedure
A total of 162 early gastric neoplastic lesions (153 patients) received an en-bloc type of ESD resection. Table 1 provides a summarized baseline information of recruited patients including frequency distribution and their corresponding clinical parameters. The rate of completeness of resection and NEC outcome were 95% and 22.2%, respectively. Three fourth of the participants (74.1%) were males and the mean age (± standard deviation) of the study population was 61.10 ± 8.76 years.
Majority of the neoplastic lesions (58.6%) were located in the lower one third of the stomach and 71.6% of all the tumors had a macroscopic type belonging to the group with purely superficial elevated or depressed lesions (0-IIa or 0-IIc) and/or their corresponding mixed variants where the superficial elevated or depressed is a predominant feature (Group 2). All other macroscopic lesions such as protruding (0-I), purely superficial flat (0-IIb) and mixed variants of superficial flat constituted for 28.4% (Group 1). A large proportion of gastric lesions had a size ≤ 2 cm (68.5%, 111 lesions), absence of ulceration (83.0%, 135 lesions), and differentiated type of histology (79.6%, 129 lesions). According to Lauren’s classification of gastric tumors, a total of 129 lesions (79.6%) displayed an intestinal-type morphology whereas nearly 20% were non-intestinal (diffuse/mixed histology). In terms of depth of tumor invasion, there were higher number of intra-mucosal lesions as compared to the submucosa, 81.5% and 18.5%, respectively. The horizontal margin was positive in three lesions (1.9%), and four lesions (2.5%) had positive vertical margin. Positive lympho-vascular invasion was identified in three lesions (1.9%).
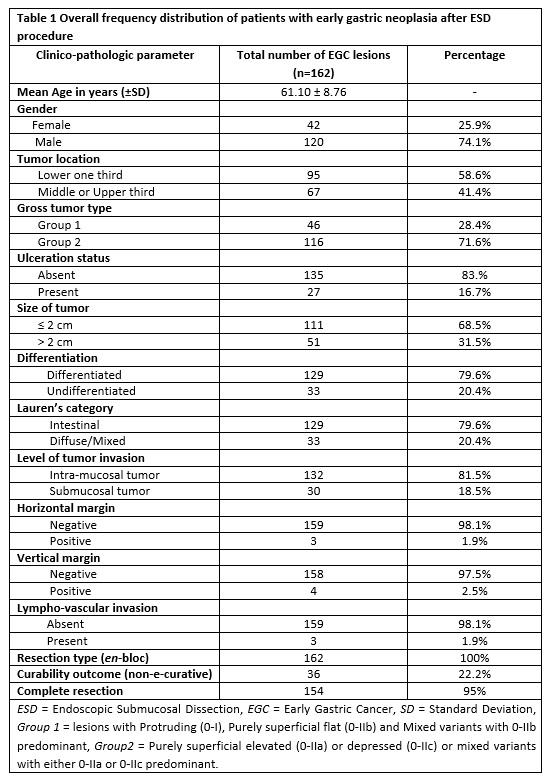
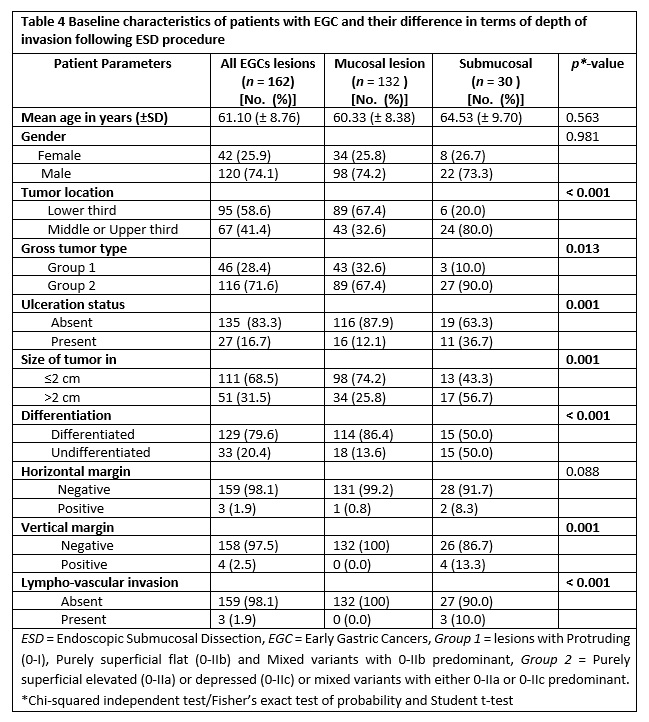
The baseline characteristics of patients with regard to depth of tumor invasion (mucosal vs submucosal) are shown in table 4. Out of 162 lesions, 18.5% (30 lesions) had invasion into the submucosal layer. Considering tumor location and macroscopic endoscopic features, tumors in the upper two third of stomach and those having 0-IIa/0-IIc or their mixed types with 0-IIa/0-IIc predominant had submucosal invasion of 80% and 90%, correspondingly. In terms of histologic differentiation, the number of cases in both differentiated and undifferentiated groups exhibiting submucosal invasion were equal (50% each).
Factors that contributed to NEC resection after ESD procedure
Table 2 summarizes the demographic and clinico-pathologic factors of patients with early gastric neoplasia in relation to the post-ESD curability outcome. Accordingly, all patients were divided into two groups (e-curative vs non-e-curative). Overall, there were 126 early gastric lesions (77.8%) that received curative ESD resection whereas 36 lesions had a NEC resection (22.2%). We tried to compare e-curative and NEC groups in terms of their demographic and clinico-pathologic features using a Chi-squared independent test or Fisher’s exact test and student t-test applied as appropriate. Hence, there was significant difference in curability outcome among tumor-associated factors such as tumor location, differentiation, size of tumor, and depth of tumor invasion (all with p < 0.001). Likewise, a significant difference was found between these groups of patients with regard to macroscopic tumor type (p = 0.002), ulcerative finding (p = 0.011), horizontal margin (p = 0.011), vertical margin (p = 0.002) and lympho-vascular invasion (p = 0.010) following endoscopic resection. On the contrary, no significant difference was seen with respect to gender of patients (p = 0.472) and the difference in age was only marginally significant (p = 0.046).
Subsequently, the predictive impact of all clinico-pathologic factors of enrolled patients with early gastric cancer following ESD procedure was investigated using logistic regression analysis as depicted in table 3.
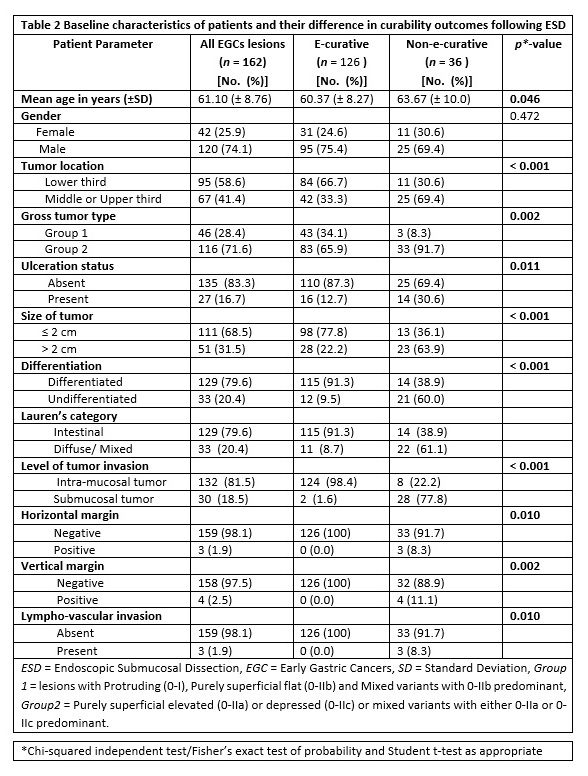
Initially, the role of each tumor-associated variable was examined separately for curability outcome in a univariate analysis. As a result, significant effect on non-curability was obtained in association with tumors located in the upper two third, larger tumor size (> 2 cm), undifferentiated histology and tumors that belonged to diffuse or mixed by Lauren’s classification (all with p < 0.001). In addition, univariate analysis revealed significant associations with NEC resection when lesions; were ulcerated (p = 0.001), had purely superficial elevated or depressed or their mixed counterparts (p = 0.006), and when patients had advanced age (p = 0.049).
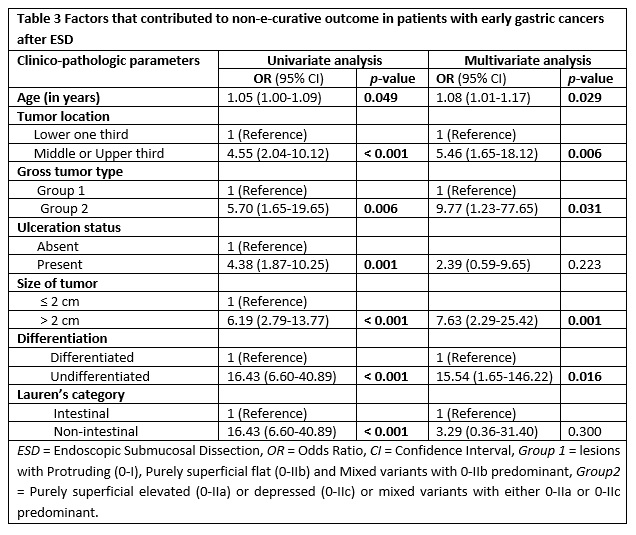
Finally, a multivariate logistic regression analysis was carried out (table 3) in which all the clinico-pathologic factors were considered together in order to further investigate factors that independently predict a NEC outcome. Thus, tumor location in the upper two third of stomach (odds ratio [OR] 5.46, confidence interval [CI] 95%, 1.65-18.12, p = 0.006), tumor size greater than 2 cm (OR 7.63, CI 95%, 2.29-25.42, p = 0.001), microscopically undifferentiated tumor (OR 15.54, CI 95%, 1.65-146.22, p = 0.001), and old age (OR 1.08, CI 95%, 1.01-1.17, p = 0.029) were found to have independent predictive influence on NEC resection of early gastric tumors.
In a similar fashion, purely superficial elevated or depressed or mixed gross type lesions with either elevated or depressed component predominating, independently contributed to a NEC outcome (OR 9.77, CI 95%, 1.23-77.65, p = 0.031). The presence of ulceration (OR 2.39, 95% CI, 0.59-9.65, p = 0.223) and non-intestinal (diffuse or mixed) tumors by Lauren’s classification (OR 3.29, 95% CI, 0.36-31.40, p = 0.300) did not show significant contribution to no-curability after multivariate analysis.
Factors associated with submucosal invasion in patients with EGC after ESD
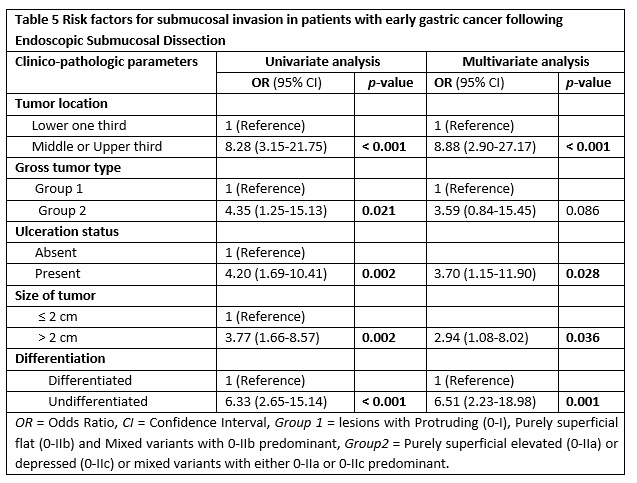
Herein, we conducted both comparative and multiple logistic regression analysis of clinico-pathologic factors with respect to the depth of invasion as illustrated in table 4 and table 5, respectively. Accordingly, significant difference was obtained in tumors located in the upper two third vs lower third (p < 0.001), macroscopic type of 0-IIa/0-IIc or their mixed types with 0-IIa/0-IIc predominant gross type vs 0-I/0-IIb and mixed with 0-IIb dominant (p = 0.013), lesions with ulcerative finding vs non-ulcerated (p = 0.001), size ≥ 2 cm vs size < 2 cm (p = 0.001) and undifferentiated vs differentiated histology (p < 0.001).
Subsequently, factors that showed significant difference were analyzed using univariate and multivariate logistic regression (table 5). In a univariate analysis, submucosal infiltration was significantly related with tumors of upper two third location, gross type 0-IIa/0-IIc or their mixed types with 0-IIa/0-IIc predominant, ulcerated lesions, lesions with ≥ 2 cm size and undifferentiated histology (all with p < 0.05).
A multivariate logistic regression analysis revealed that the following tumor-related factors were independent predictors of submucosal invasion; location in the upper two third of stomach (Odds ratio [OR] 8.88, Confidence interval [CI]) 95%, 2.90-27.17, p < 0.001), ulcerated lesions (OR 3.70, CI 95%, 1.15-11.90, p = 0.028), lesions with greater than 2 cm (OR 2.94, CI 95%, 1.08-8.02, p = 0.036) and those with poor differentiation (OR 6.51, CI 95%, 2.23-18.98, p = 0.001).
Results of gastrectomy specimens for patients who had NEC outcome following ESD
Parallel to evaluation of ESD resected specimens, we tried to look at some of the gastrectomy specimens together with their lymph node dissections for patients who had a NEC with an attempt to determine the presence of residual tumor and lymph node metastasis (Figure 1). Hence, out of the total 36 lesions that satisfied the criteria for NEC outcome, 17 patients received additional surgical treatment. Fifteen of those patients who received additional surgery showed evidence of mucosal defect with chronic ulcerative and inflammatory changes or/and granulation tissue. But not cancerous cells were detected in the resected lymph nodes and surrounding tissues. Residual cancer was found in two patients who underwent surgery but none of them exhibited metastatic disease in the dissected lymph nodes.




