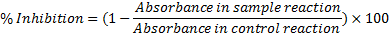ZCE and its bioactive fraction are neurotoxic and retards mosquito larval development in a dose-dependent manner
Previous studies have demonstrated inhibition of An. gambiae, Ae. aegypti, Culex pipiens, and Ae. albopictus mosquito larval development by Zanthoxylum plant extracts [39–43]. Currently, we report that mosquito larvae exposed transiently to ZCE at 15-25 ppm were immobilized and less active within the first 20 min for both An. gambiae and Ae. aegypti (data not shown). This exposure elicited 60-100% larval mortalities at LC50 9.00 ppm (95% CI 8.62-21.50) and LC50 12.26 ppm (95% CI 9.04-19.80) against An. gambiae and Ae. aegypti, respectively, within 24-h post-treatment (Table 1, Fig. 1). When compared to the negative controls that achieved 100% survival, ZCE significantly reduced larval survival rates (An. gambiae, F(5,24) = 105.5, p < 0.001; Ae. aegypti, F(5,24) = 314.4, p < 0.001) but slightly higher doses were required to match the activity of neem oil that was included as positive control. However, the variation in susceptibility to ZCE toxicity was insignificant (F(1,48) = 0.836, p = 0.365). Fractionation of ZCE yielded a bioactive hexane-soluble fraction (ZCFr.5) with similar activity to ZCE but at lower doses of LC50 1.58 ppm (95% CI 0.62-2.87) and 3.21 ppm (95% CI 2.99-3.83) against An. gambiae and Ae. aegypti, respectively (Table 1, Fig. 1). Other solvent fractions yielded no activity (Supplementary Table S1).
Table 1. Toxicity of ZCE extracts on An. gambiae and Ae. aegypti larvae 24-h post exposure.
|
Treatments
|
Concentration (ppm)
|
Larval mortality
(% mean ± S.D)
|
LC50 (95% CI)
|
|
An. gambiae
|
Ae. aegypti
|
An. gambiae
|
Ae. Aegypti
|
|
MeOH extract
(ZCE)
|
25
|
100 ± 0.00a
|
100 ± 0.00a
|
9.00 (8.62-21.502)
|
12.26 (9.04-19.799)
|
|
20
|
94 ± 1.64‡
|
79 ± 2.280a
|
|
|
|
15
|
78 ± 3.36‡
|
62 ± 1.140a
|
|
|
|
10
|
66 ± 1.82‡
|
37 ± 1.643a
|
|
|
|
5
|
38 ± 3.36a
|
2 ± 0.894a
|
|
|
|
Hexane soluble fraction (ZCFr.5)
|
10
|
100 ± 0.00a
|
100 ± 0.00‡
|
1.58 (0.62-2.87)
|
3.21(2.99-3.83)
|
|
7.5
|
94 ± 2.28a
|
87 ± 0.447‡
|
|
|
|
5
|
90 ± 1.12a
|
75 ± 2.387a
|
|
|
|
2.5
|
78 ± 0.98a
|
24 ± 3.162a
|
|
|
|
1
|
25 ± 2.23a
|
10 ± 1.673a
|
|
|
|
Neem oil (positive control)
|
3.8
|
98 ± 1.59
|
100 ± 0.00
|
|
|
Late third instar mosquito larvae of An. gambiae and Ae. aegypti were exposed to various doses of ZCE (5-25 ppm) and its active fraction (1-10 ppm) and mortalities in each setup recorded for 24-h against the controls. Mosquito population size (N) per treatment dose per replicate was 125 larvae (n = 2 biological replicates). Mean values followed by a double dagger (‡) are not significantly different. Values followed by a superscript letter are significantly different (p < 0.05, One-way ANOVA – Tukey-Kramer’s multiple comparison test). ppm denotes parts per million, S.D – standard deviation, CI – confidence interval, LC50 – lethal concentration on 50% of test population.
We also evaluated the residual effect of long term exposure on developmental growth progression. Upon treatment with sublethal dosage of either the ZCE ( or hexane-soluble bioactive fraction (ZCFr.5), the larval transition to pupal stages was abnormally extended (averagely 3.75 ± 0.1732 days, Table 2) inducing retardant phenotypes (ZCE: An. gambiae t = 15.963, p = 0.0004243; Ae. aegypti t = 23.883, p = 0.0004457; ZCFr.5: An. gambiae t = 30.311, p = 0.001087, Ae. aegypti t = 7.0162, p = 0.01272). Further, unlike the controls that achieved > 82% pupation (Table 2), pupation rate for the treated cohorts was significantly affected with many dying at larval state (ZCE: An. gambiae t = -18.826, p = 0.0007721; Ae. aegypti t = -19.078, p < 0.0001; ZCFr.5: An. gambiae t = -14.233, p = 0.0001549, Ae. aegypti t = -31.787, p = 0.0006135). Overall, species-specific perturbations were noted, with more treatment effects in An. gambiae than Ae. aegypti (ZCE, t = -11.539, p = 0.0005719; ZCFr.5, t = -6.3535, p = 0.02296).
Table 2. Developmental duration of mosquito larvae treated with sublethal doses of Z. chalybeum extracts
|
Treatment
|
Pupation time (days)‡
|
Pupation rate (%)
|
|
An. gambiae
|
|
Non-treated control
0.01% ethanol control
ZCE
ZCFr.5
|
5.30 ± 0.17a
5.60 ± 0.20a
9.10 ± 0.00b
9.40 ± 0.36b
|
85.33 ± 3.10a
82.61 ± 3.89a
37.22 ± 1.51b
34.73 ± 1.72b
|
|
Ae. aegypti
|
|
Non-treated control
0.01% ethanol control
ZCE
ZCFr.5
|
4.80 ± 0.35c
5.00 ± 0.26c
8.90 ± 0.10d
8.80 ± 0.90d
|
89.40 ± 2.71c
86.15 ± 1.90c
54.65 ± 2.14d
50.55 ± 0.39d
|
‡Time period recorded from early L3 instar until pupal transformation for three replicates. Values presented as means ± standard deviation (S.D). Mean values followed by similar superscript letters are not significantly different (One-way ANOVA – Tukey-Kramer’s multiple comparison test, p < 0.05). Sublethal dosages used: ZCE: 8 ppm, 10 ppm; ZCFr.5: 1.0 ppm, 3 ppm for An. gambiae, and Ae. aegypti, respectively
Phytochemical analysis identified an abundance of insect growth reducing compounds
To identify the chemical constituents present in the bioactive hexane-soluble fraction (ZCFr.5), the yellow brown oily sample was subjected to a gas chromatography-mass spectrometer (GC-MS) analyzer. From a total of 67 components (Supplementary Table S2), the GC-MS analyzer detected among other compounds the presence and abundance of previously reported insect growth-reducing agents[42],[44], corresponding to 2-tridecanone (1) (Rt 18.2267), hexadecanoic acid (2) (Rt 23.3864), (Z,Z)-9,12-octadecadienoic acid (linoleic acid) (3) (Rt 25.0829), sesamin (1,3-Benzodioxole,5,5'-(tetrahydro-1H,3H-furo[3,4-c]furan-1,4-diyl)bis-,[1S-(1.alpha.,3a.alpha.,4.beta.,6a.alpha.)]-) (4) (Rt 34.3786) and β-caryophyllene (5) (Rt 38.38)(Fig. 2A, 2C). Proportionately, the compounds 1, 2, 3, 4, and 5 contributed to 10.82%, 7.71%, 11.14%, 11.43%, and 10.38%, respectively, representing 51.48% of the total active biomass (Fig. 2B).
Perturbed acetylcholinesterase (AChE) activity linked to larval lethality
Transient exposure of mosquito larvae to ZCE and its bioactive fraction for <5 h exhibited no obvious morphological aberrations but drastically reduced larval swimming behavior, induced muscle paralysis and immobilization leading to death. These observations are closely linked to neuromuscular toxicity effects reported by Tomé et al.[45]. To ascertain whether the observed acute toxicity on mosquito larvae was as a result of suppressive effects of ZCE and its bioactive fraction on AChE, we performed the biochemical colorimetric Ellman’s enzymatic assay and RT-qPCR gene expression analysis. Due to the low amount of ZCFr.5, we only assayed ZCE for larval AChE activity inhibition. ZCE exhibited dose-dependent AChE inhibitory effect in mosquito larvae (Fig. 3B), attaining IC50 136.0 µg/mL (95% CI 111.3-547.3) and 277.5 µg/mL (95% CI 47.4-293.4) for Ae. aegypti and An. gambiae, respectively. The AChE inhibition achieved by ZCE was preferably 1.4-2.73 fold higher in Ae. aegypti (44.19-81%) than An. gambiae (16.11-59.59%). Irrespective of ZCE dosage, the toxicity susceptibility of AChE significantly varied between Ae. aegypti and An. gambiae (Welch two-sample t-test, p < 0.001), highlighting a species-specific response. Relative to propoxur, a specific carbamate-based irreversible AChE inhibitor, the mean ZCE activity varied significantly between the species (An. gambiae; t-test, t= -8.4174, df = 4, p = 0.001055; Ae. aegypti; t=-2.9963, df = 4, p = 0.03648). Agreeably, the larval treatment with ZCFr.5 was associated with 0.0764 and 0.017-fold Ace transcriptional changes in Ae. aegypti and An. gambiae, respectively (Fig. 3A), but these expressions were not significantly different from each other (t-test = 2.4406, df = 4, p = 0.066). Meaningful and significant (p > 0.05) negative correlation coefficients between the mean LC50 and AChE inhibition values of ZCE were obtained, but not varied between the two mosquito species (Supplementary Table S3).
ZCFr.5 modulates mosquito larval ecdysteroid biosynthetic and transcriptional regulatory genes
Lignan-based prenylpropanoid and 2-tridecanone compounds are believed to inhibit ecdysteroids and their biosynthetic-dependent cytochrome P450 activities[44],[46], interfering with insect growth. As expected, the sesamin-rich ZCE root fraction altered the transcriptional expression of ecdysteroid biosynthetic Halloween and regulatory genes (Fig. 4). Notably, the larvicide treatment significantly down-regulated the expression of larval ‘black box’ cytochrome P450 spook (Cyp307a1), βFtz-F1, disembodied (Cyp302a1), and ecdysteroid receptor (EcR) genes. We further noted that, with an exception of Cyp302a1 in Ae. aegypti, the gene expressions of Cyp314a1, Cyp306a1, and dHAT were not significant from each other (t-test, p > 0.05; Fig. 4A-B), but remarkably modulated relative to the controls. Intriguingly, while juvenile hormone (JH) expression levels decrease during the last larval instar to allow 20E-induced transformation into pupal stages, we established that the expression of JH biosynthetic rate-limiting enzyme, JH acid O-methyltransferase (JHAMT), in ZCFr.5-treated larvae remained relatively higher compared to that of controls (Fig. 4C-D). This finding could suggest low levels of circulating hemolymph 20E to suppress JH and further underscoring the observed larval growth retardations and precocious pupations. These findings confirmed that indeed the larval treatment that targeted AChE activity also perturbed ecdysteroidogenic pathway associated genes delaying larval-pupal transitions and inducing retardant phenotypes.